This site is intended only for the use of UK Healthcare Professionals
SMA® Althéra®
- Extensively hydrolysed, hypoallergenic, whey-based formula (eHF) for the effective first line dietary management of the majority (~90%) of infants with cows’ milk allergy (CMA) and other conditions where an extensively hydrolysed formula is indicated1–4 in formula fed babies.
- The only whey-based eHF that is both Halal and Kosher certified.
- Althéra is the only eHF to show similar efficacy* to an amino acid formula**(AAF). eHFs are not recommended where only an AAF is indicated.1,2
- Supports normal growth and development1,2
- Preferred taste over other casein based eHF which may help feeding acceptance, tolerance and health economic savings2,6-8
- Nutritionally complete. Suitable as a sole source of nutrition from birth or supplementary feeding from 6 months of age
*In a RCT
**in mild-moderate cows’ milk allergy (CMA)
*This is for professional evaluation use only
Lactose (milk), maltodextrin, vegetable oils (sunflower, rapeseed, coconut), hydrolysed whey protein (milk), minerals (calcium glycerophosphate, potassium phosphate, magnesium chloride, calcium chloride, sodium chloride, potassium chloride, ferrous sulphate, sodium phosphate, zinc sulphate, potassium citrate, copper sulphate, manganese sulphate, potassium iodide, sodium selenate), emulsifier (E472c), Mortierella alpina oil (ARA), oil from the microalgae Schizochytrium sp. (DHA), choline bitartrate, acidity regulator (E 330), vitamins (C, E, niacin, pantothenic acid, riboflavin, A, thiamin, B6, folic acid, K, D, biotin, B12), L-arginine, L-histidine, taurine, inositol, L-carnitine.
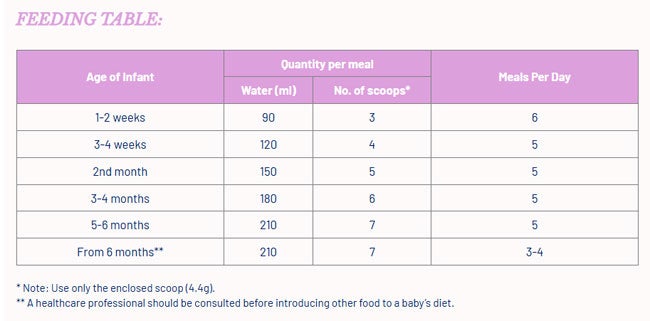
- 400g tin (with a 4.4g scoop).
- Use under strict medical supervision after full considerations of all the feeding options available, including breastfeeding.
- For oral or enteral use only.
- Not suitable for galactosemia, glucose-galactose malabsorption, lactose intolerance
- Althéra® is not recommended where only an amino acid formula is indicated.
Do not add any extra powder or water to make feeds stronger or weaker. Using too much or too little powder can make a baby ill. We recommend preparing each feed in individual bottles when required, feed immediately. If there is any unfinished infant milk left after feeding, throw it away. Always hold the baby while feeding. Never leave the baby alone during feeding as there is a risk they might choke.
Contains milk.

Shelf life of 24 months from date of manufacture when stored at room temperature.
Once opened, reseal & store in a cool dry place and use within 3 weeks.
ACBS approved, prescribable on FP10 (GP10 in Scotland). Suitable as a sole source of nutrition from birth or supplementary feeding from 6 months and up to 3 years of age. For the dietary management of infants with cows’ milk allergy
Nestlé Health Science produces a range of foods for special medical purposes (FSMP) for use under medical supervision.
We believe that breastfeeding is the ideal nutritional start for babies as breast milk provides a balanced diet and protection against illness for a baby. We fully support the World Health Organisation’s recommendation of exclusive breastfeeding for the first six months of life followed by the introduction of adequate nutritious complementary foods along with sustained breastfeeding up to two years of age. We also recognise that breastfeeding may not be an option due to certain medical conditions. Parents should only feed infant formula for special medical purposes under supervision of a healthcare professional after full consideration of all feeding options, including breastfeeding. Continued use has to be assessed by the healthcare professional in relation to the baby’s progress bearing in mind any social and financial implications for the family. Infant formula should always be prepared, used and stored as instructed on the label in order to avoid risks to a baby’s health. Food for special medical purposes intended for infants must be used under medical supervision. For oral/ enteral use only. Suitable as a sole source of nutrition from birth or supplementary feeding from 6 months and up to 3 years of age.
