This site is intended only for the use of UK Healthcare Professionals
Modulen® IBD Powder
Modulen® IBD is a nutritionally complete, Food for Special Medical Purposes for the dietary management of Crohn’s disease.
Modulen® IBD is a whole protein powdered formulation for use as a sole source of nutrition during the active phase of Crohn’s disease and for nutritional support during the remission phase. It is 100% casein based and provides 25% of the fat as medium chain triglycerides (MCT).
*This is for professional evaluation use only
Glucose syrup, milk protein, sucrose, milk fat, medium chain triglycerides (MCT) oil, minerals (magnesium chloride, calcium phosphate, sodium citrate, potassium citrate, potassium hydroxide, calcium carbonate, potassium chloride, manganese sulphate, ferrous sulphate, zinc sulphate, copper sulphate, sodium molybdate, chromium chloride, potassium iodide, sodium selenate), corn oil, emulsifier (soy lecithin), vitamins (C, E, niacin, pantothenic acid, B6, thiamin, A, riboflavin, folic acid, K, biotin, D, B12), choline bitartrate.
Halal certified and Gluten free.
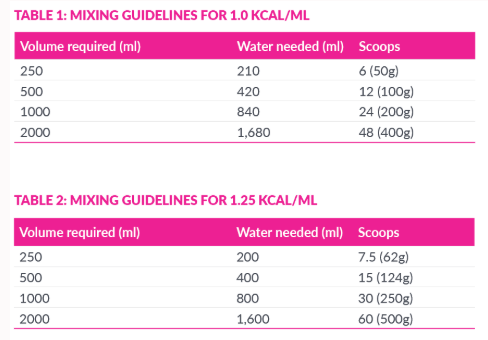
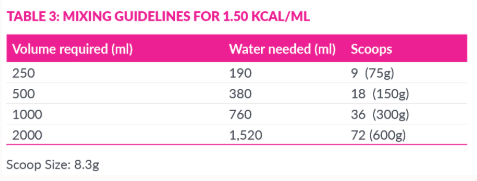
One 350g Modulen® IBD tin provides 1750 kcl. The dosage of Modulen® IBD is dependent on the age, weight and clinical condition of the patient. The recommended feed concentration is 1kcl/ml (20%). As a guide, Modulen® IBD can also be concentrated to provide 125 kcal/ml (25% concentration) or 1.5 kcal/ml (30% concentration) but additional fluid should be advised.
- Wash hands thoroughly. Follow the mixing table (see below) and select the volume required.
- Measure cool boiled or bottled water (room temperature) and pour into a clean bowl or container.
- Scoop and level the desired amount of powder using the scoop in the tin or weigh in grams.
- Add the powder to the water and immediately stir until well mixed.
- After use, store the scoop inside the can.
Shelf life of 24 months from date of manufacture when stored at room temperature. Consume the contents within 4 weeks of opening. Once reconstituted, use within 4 hours at room temperature or 24 hours if refrigerated.
Indications
ACBS approved, prescribable on FP10 (GP10 in Scotland). Indicated for the dietary management of Crohn’s disease. For use as a sole source of nutrition during the active phase of Crohn’s disease or nutritional support during the remission phase in malnourished patients.
Contraindications
Not suitable for use in cows’ milk protein allergy.
Precautions
For oral or enteral feeding only. Only suitable for those over 5 years. Do not add food or drugs, if product is tube fed.
Modulen® IBD is a Food for Special Medical Purposes for the dietary management of Crohn’s disease. Must be used under medical supervision. Suitable for use as the sole source of nutrition.
